Hybrid hybrid car
28 December 2008 - News
We are familiar with hybrid cars that use both hydrocarbon fuel and a battery or a fuel cell for propulsion. But why not have one with a hybrid fuel cell system as well? A group of researchers from Queen's University explain how (Wechsler et al. 2008 DOI).
They first observe that the current contenders in fuel cells based on hydrogen storage either react in a exothermic reaction e.g. boron hydrides and metal hydrides or in a endothermic reaction e.g. cycloalkanes and piperidines (at the expense of fuel cell efficiency) and then propose that in a hybrid system heat generated by one reaction can be used to drive the other one. The symbiosis does not end there: the exotherms are generally solids but can be dissolved in liquid and pump-able endotherms.
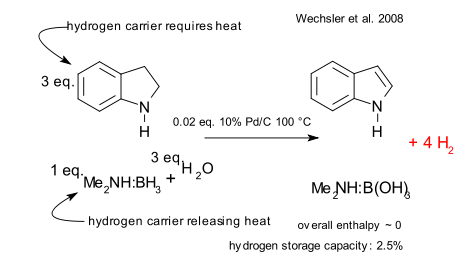
In one demonstration endothermically reacting indoline is mixed with the exothermically reacting dimethylamine.borane (think ammonia borane) and water mixture with as catalyst palladium on carbon: both reactions go to completion without a nett enthalpic change. The calculated hydrogen storage capacity is 2.5%. The researchers are honest about the limitations of the system: the borate formed in the reaction is not soluble and the rates of both reactions are dissimilar.
