Double metallocenes
22 November 2008 - Basics
Double metallocenes are a class of organometallic compounds in which the cyclopentadienyl ligand in a Cp2M metallocene is replaced by a pentalenyl ligand (review DOI). They are of some importance as catalysts in olefin polymerization.
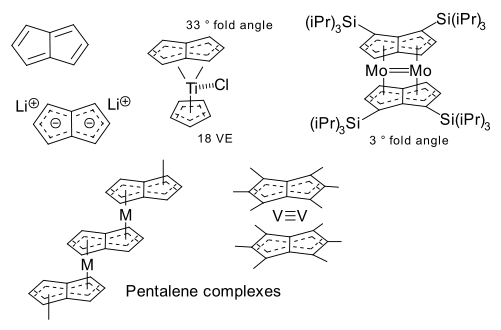
The first such compound was prepared by Jonas et al in 1997 (DOI). Reaction of titanocene dichloride with the dilithium salt of pentalene forms Ti(eta8-C8H6)(Cp) with titanium simultaneously coordinating to 8 positions on the pentalene frame and 5 on the Cp frame. The related Ti(Pn)(Cp)Cl is obtained by further reaction with dichloromethane as an 18 VE compound. Interestingly, although the pentalenyl ligands are aromatic, they are not planar but have a fold angle in the range of 30 - 40°.
The bimetallic bis-pentalene sandwich compound Mo2(C8H4(1,4-Si(iPr)3)2)2 was prepared by Cloke et al. in 1998 (DOI) by reaction of K2(C8H4(1,4-Si(iPr)3)2) and molybdenum acetate. In this compound the fold angle has vanished and the two molybdenum centers form a double bond.
When the bimetallic orientation is trans rather than cis it is in principle possible to create (M(Pn))n polymers which are of potential interest as molecular wires. One of the first efforts to be recorded , a (*Cp)2PnFe2 trimer, is one by Manriquez et al. in 1988 (DOI).
Most recently the double metallocene family was extended by O'Hare et al. with a range of *Pn2M2 compounds with M vanadium through nickel and *Pn the pentalene version of pentamethylcyclopentadiene (DOI). Intriguingly the iron sandwich resists synthesis with the pentalene ligands instead opting for dimerization.
