White phosporus tamed
02 July 2009 - Part II
White phosphorus (P4) is known to react violently with oxygen and Mal et al. (DOI) have found a novel way to keep this from happening by locking the molecule into an iron cage.
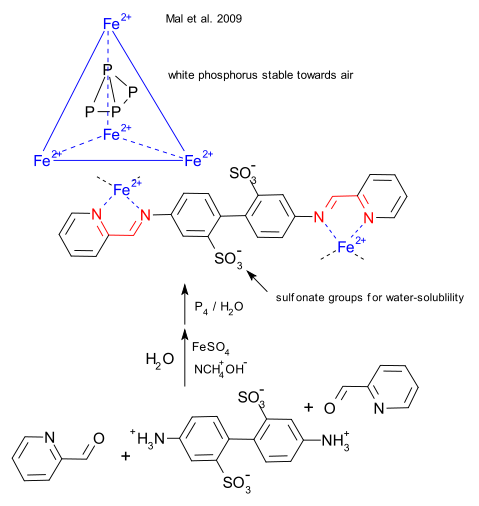
This cage is prepared in water from iron sulfate , 4,4-diaminobiphenyl-2,2-disulfonic acid, 2-formylpyridine and tetramethylammonium hydroxide as phase-transfer catalyst. The hydrocarbon components condense to an extended molecule and 5 of them form the vertices of a tetrahedron which is held together by a self-assembly process as the terminal diimine groups (in red) coordinate to iron. (Mail et al. DOI). When exposed to phosporus, the water-soluble host-guest complex P4@Fe4 forms. The pore-size of 1 angstrom does not prohibit oxygen from entering the cage, rather it is theorized that any oxidation product would simply be too large to fit. Added benzene replaces the phosphorus and the process is reversible. The only disadvantage is that the phosphorus capacity of the container is only 4%.
See also: white phosphorus tamed part I
