When calcium met nitrogen
17 March 2021 - Inorganic chemistry
This is an interesting research theme. Reduce an alkaline earth metal to it's plus one oxidation state and watch what happens next. End of last year there was Cameron Jones exploiting magnesium dimer L-Mg-Mg-L with each magnesium a reactive plus one (blog) and there was also from the boron group Philip Power with a plus one R-Al aluminium compound (blog).
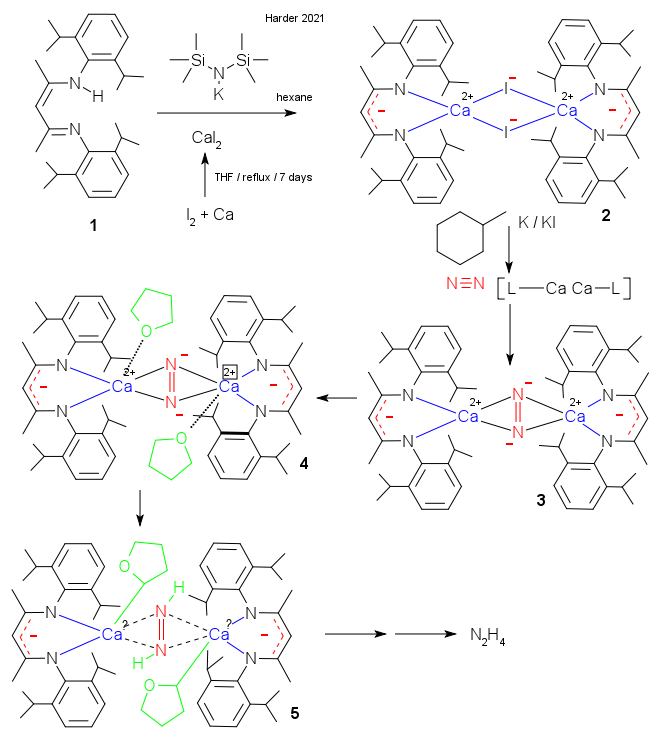
The Frenking / Harder groups have now set their sights at calcium. They report their attempt to create a L-Ca-Ca-L dimer in a similar way as Jones where L is a beta-diketiminate ligand (Rösch et al. DOI). The outcome was unexpected. The dimer was predicted to materialize by reducing the corresponding L-Ca iodine dimer but whatever monster came out was just too reactive and escaped isolation.
The authors explain that instead it takes on whatever is available. With potassium metal as reducing agent it rather reacts with solvent benzene or any other aromatic solvent. More surprisingly, in absence of an aromatic solvent (all-aliphatic methylcyclohexane) and with potassium dispersed on potassium iodide a complex with nitrogen is formed from the "protective" nitrogen blanket.
The compound can be stabilized with tetrahydrofuran and the crystal structure confirms de nitrogen unit has a double bond and is a dianion. Heating this compound to 60 degrees results in alpha hydrogen abstraction from the THF ligand with the hydrogen atom ending up with nitrogen and the complex then breaks down into ethylene, the acetaldehyde calcium enolate and diazene. The latter byproduct eventually forms hydrazine. That almost sounds like nitrogen reduction but calcium is of course not doing a real catalytic Haber-Bosch reaction (HB).
In traditional HB a transition metal can use partially filled d-orbitals to interact with empty pi-star orbitals at nitrogen in a concept called the Dewar-Chatt-Duncanson model. The authors propose that calcium mimicks the transition metal by doing the very same. But d-orbital participation in an alkaline metal is controversial! In 2019 Frenking unveiled calcium octacarbonyl (see blog here) but his suggestion in 2019 that this compound owes its existence to pi backbonding was met with criticism and an angry article (DOI) In the new work Frenking did the calculations and sticks to his guns.
The authors slyly remark that although the calcium dimer did not materialize, the new nitrogen adduct is a strong reducing agent by itself by ejecting nitrogen in addition to the electrons and even stronger than the aforementioned magnesium dimer. The Jones lab is notified.