The next Umemoto
22 May 2021 - Precision synthesis
 Speaking of late-stage functionalization (LSF) in organic chemistry (see previous blog) it is what the list of research interests of the Tobias Ritter group heads of with. In a recent publication Ritter presents (Jia et al.DOI) a new reagent that can be used to add trifluoromethyl groups, although in fairness LSF is not mentioned. The new compound is a variation on the well-known Umemoto reagent (5-(Trifluoromethyl)dibenzothiophenium trifluoromethanesulfonate) used in trifluoromethylation reactions.
Speaking of late-stage functionalization (LSF) in organic chemistry (see previous blog) it is what the list of research interests of the Tobias Ritter group heads of with. In a recent publication Ritter presents (Jia et al.DOI) a new reagent that can be used to add trifluoromethyl groups, although in fairness LSF is not mentioned. The new compound is a variation on the well-known Umemoto reagent (5-(Trifluoromethyl)dibenzothiophenium trifluoromethanesulfonate) used in trifluoromethylation reactions.
Where synthesis of the Umemoto reagent takes several synthetic steps described as difficult, the new reagent trifluoromethyl thianthrenium triflate (TTT) is synthesized in one simple step from thianthrene and triflic anhydride. The first mechanistic step in this reaction is a nucleophilic addition and there appear to be two main driving forces for the ease of the reaction (supported by radical trapping experiments and ESR), scission of the inherently weak sulfur sulfur bond to a radical / radical-ion pair followed by expulsion of sulfur dioxide allowing two radical fragment to unite. The difference with the Umemoto reagent (with a dibenzothiophene core) is just one methylene group but Ritter explains dibenzothiophene will resist reaction with the anhydride.
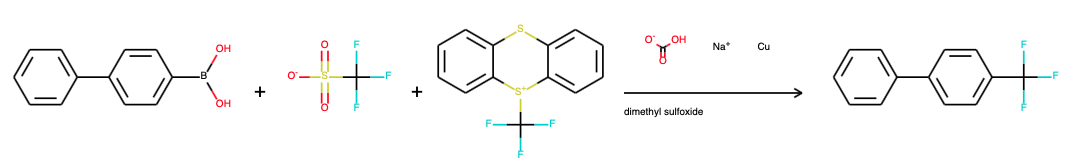
TTT is surprisingly stable, it only has to stored away from light and TTT has a diverse reactivity. It reacts with copper and a boronic acid in a cross-coupling reaction to the trifluoromethyl aryl as an electrophilic reagent. Under a blue LED light it can also react in radical trifluoromethylation for example in reaction with 2,6-dimethoxypyridine. With sodium trimethylsilanolate TTT is converted to a nucleophilic reagent and reacts for example with the keto group on 2,2,2-trifluoroacetophenone.
Returning to the late stage functionalization theme, examples described in the article are the trifluorination of caffiene, rotenone and racecadotril.
