Remdesivir latest
08 February 2020 - Antipandemics
 With large parts of China in a lock down on account of the Wuhan corona virus the hunt is on for a defense, a treatment or a cure. A lot of attention is directed at vaccines as in regular flu but with development work, testing and production a working vaccine in the hands of medics can be months away. Another potential avenue is a synthetic antiviral. It is not that we are lacking them, examples of commercially available drugs are Baloxavir marboxil, Peramivir, and Zanamivir. The 2003 SARS pandemic eventually petered out and has been treated with antivirals in general as yet no vaccine exists. In the 2005 H5N1 Pandemic the antiviral Oseltamivir (Tamiflu) became a household name. Governments were stockpiling it but years later it was determined Tamiflu is not that efficient. Middle East respiratory syndrome (MERS) was first diagnosed in 2012 and never went away with new cases reported every year. Same story here: no vaccine and no virus-killer wonder drug.
With large parts of China in a lock down on account of the Wuhan corona virus the hunt is on for a defense, a treatment or a cure. A lot of attention is directed at vaccines as in regular flu but with development work, testing and production a working vaccine in the hands of medics can be months away. Another potential avenue is a synthetic antiviral. It is not that we are lacking them, examples of commercially available drugs are Baloxavir marboxil, Peramivir, and Zanamivir. The 2003 SARS pandemic eventually petered out and has been treated with antivirals in general as yet no vaccine exists. In the 2005 H5N1 Pandemic the antiviral Oseltamivir (Tamiflu) became a household name. Governments were stockpiling it but years later it was determined Tamiflu is not that efficient. Middle East respiratory syndrome (MERS) was first diagnosed in 2012 and never went away with new cases reported every year. Same story here: no vaccine and no virus-killer wonder drug.
A compound making the rounds in 2020 China is remdesivir, an existing antiviral drug that has already proven to be effective against Ebola and Marburg. Remdesivir has been successfully tried on a patient in the US as reported in the New England Journal of Medicine on 31 January (DOI).
In a letter to Nature on 4 February a group of researchers from Wuhan (Wuhan Institute of Virology) and Beijing (National Engineering Research Center for the Emergency Drug) report that remdesivir and chloroquine are effective in vitro. They infected Vero cells with the isolated corona virus and measured cytotoxicity in a cell counter kit and the proliferation of new viruses with RT-PCR. They were also able to visualise the viruses in immunofluorescence by adding a so-called Goat anti-Rabbit antibody labeled with a Alexa Fluor formulation.
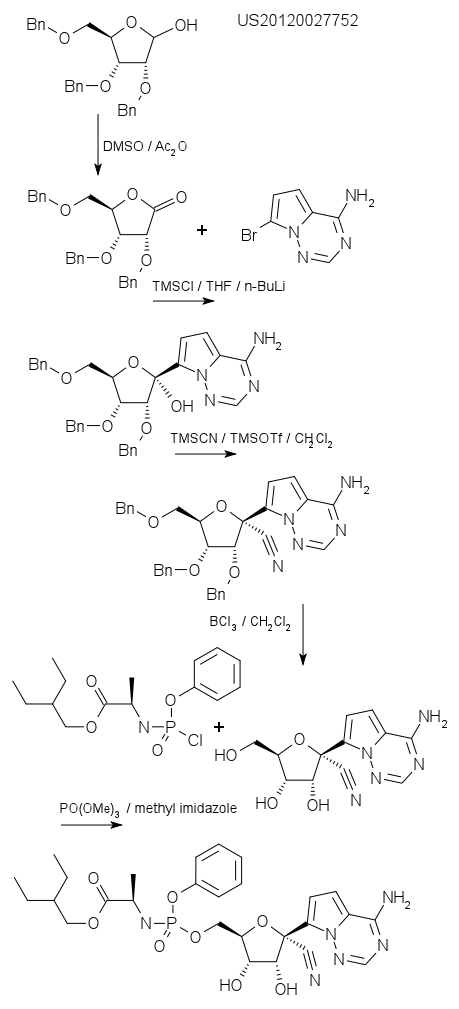
Remdesivir is a adenosine analogue, basically a purine and a ribose. The molecule can insert itself into viral RNA making the virus crash. The compound was developed by Gilead Sciences (the Oseltamivir people) years ago and a patent describing the synthesis is US73474980. Chloroquine has been around for decades and is basically an antimalarial drug. It interferes with the fusion of a virus and a cell.
Meanwhile the Wuhan researchers appear to have taken out a patent for remdesivir as an corona virus specific drug (link). This is problematic as Gilead has already patented the compound for this purpose in 2016 including in China (Link). Several days ago the company announced a clinical trial to be conducted with 270 patients in Beijing (Link) Another clinical trial announced by the president of the Chinese Academy of Medical Sciences will be held in Wuhan (Link).
Update 10 February. Earlier The Lancet study published 31-01 mentions oseltamivir (DOI) . A Nature comment mentions favipiravir, ribavirin and galidesivir and explain their attack mode (Link).
Update 13 February. New study had remdesivir tested in animals exposed to the MERS coronavirus (DOI). Research group includes Gilead people. Strange use of the word "inoculation" because the experimental monkeys were for sure infected. Remdesivir was found to be effective administered before and after the onset of infection. Chinese company BrightGene Bio-Medical Technology is reported to be ready for remdesivir production (Link). No patents but the company has patented Oseltamivir work in the past.
Update 14 February: Wired has the full story on the clinical trials taking place (link). Two antivirals intended for HIV, lopinavir and ritonavir will also be trialed. Like remdesivir they also work by messing with a key enzyme produced by the virus. No word of chloroquine trials yet.
Update 18 February: Chloroquine successfully tested in clinical trial. Report from Chinese state-owned website here and confirmation from Belgian virologist Marc Van Ranst in news report here (in Dutch).
Update 20 February: Just read in The Guardian that the WHO is not backing chloroquine? They are betting on lopinavir / ritonavir and remdesivir.
