FLP twice over.
12 July 2010 - FLP chemistry Part 6
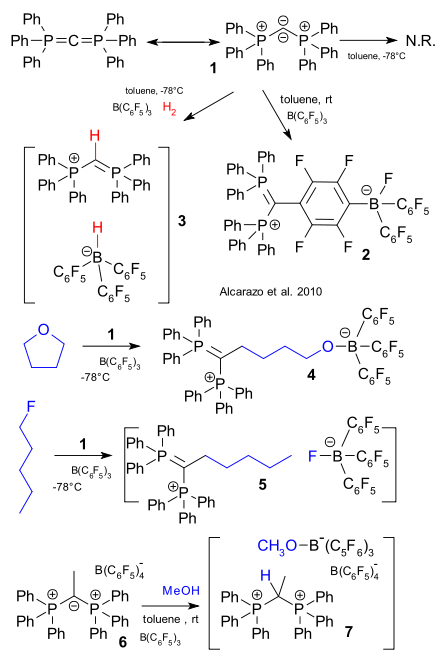 Alcarazo et al. describe a new Frustrated Lewis Pair (see FLP part V) with new Lewis acid hexaphenylcarbodiphosphorane (1) and the familiar Tris(pentafluorophenyl)boron (DOI). At room temperature the compounds react in nucleophilic substitution, at -78°C they do nothing but add hydrogen at that temperature and salt 3 forms as a result of heterolytic hydrogen cleavage. The new pair is also good at C-O bond cleavage (THF), C-F cleavage as in the fluoropentane example and in Si-H cleavage. Remarkably the pair is still active when 1 is protonated or alkylated such as in compound 6. It is not reactive enough for hydrogen but it can still cleave methanol. A frustrated Lewis pair twice over.
Alcarazo et al. describe a new Frustrated Lewis Pair (see FLP part V) with new Lewis acid hexaphenylcarbodiphosphorane (1) and the familiar Tris(pentafluorophenyl)boron (DOI). At room temperature the compounds react in nucleophilic substitution, at -78°C they do nothing but add hydrogen at that temperature and salt 3 forms as a result of heterolytic hydrogen cleavage. The new pair is also good at C-O bond cleavage (THF), C-F cleavage as in the fluoropentane example and in Si-H cleavage. Remarkably the pair is still active when 1 is protonated or alkylated such as in compound 6. It is not reactive enough for hydrogen but it can still cleave methanol. A frustrated Lewis pair twice over.
