FLP recap
26 August 2011 - Frustrated Lewis Pairs
 This is an instant frustrated Lewis pair update: for earlier episodes see here, here, here, here, here, here and here
This is an instant frustrated Lewis pair update: for earlier episodes see here, here, here, here, here, here and here
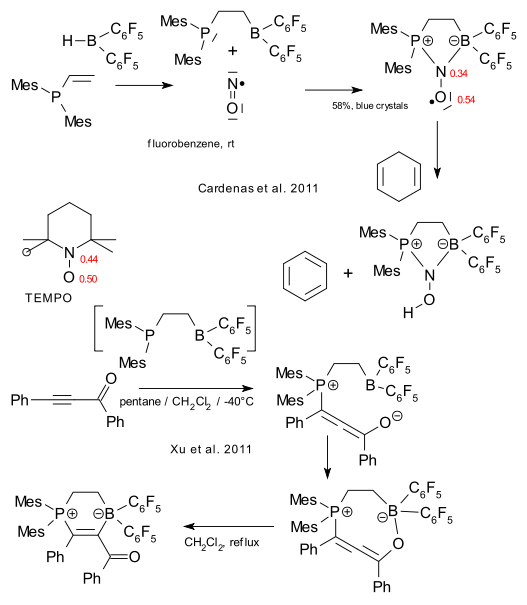
Grimme, Warren & Erker have added nitric oxide to the list of substrates for the Mes2PCH2CH2B(C6F5)2 system (Cardenas et al. 2011 DOI). The new compound is similar to TEMPO and contrary to it or regular NO itself it can abstract protons for example from cyclohexadiene to form the N-alcohol and benzene!. This effect is attributed to reduced electron density at the nitrogen atom.
The Grimme/Erker team also brings you the adduct of said P/B system with diphenylynone (Xu et al. 2011 DOI), the first reaction product a cyclic allene and then after heating it up a substituted enone. With the introduction of hydrogen the P/B system is capable of catalytic (metal free!) ynone hydrogenation.
Meanwhile the Stephan team have demonstrated imine hydrogenation using (catalytic) B(C6F5)3 and diisopropylamine as hydrogen source (Farrell et al. 2011 DOI).
Also in the recent literature: more substrates not save from the FLP treatment: dimethylpentafulvene (DOI), more FLP designs, for example a diborane ( DOI) and a zirconocene phosphinoaryloxide (DOI) and much mechanistic speculation: (DOI / DOI / DOI).
