Tetracene as drug precursor
29 June 2019 - Orgo
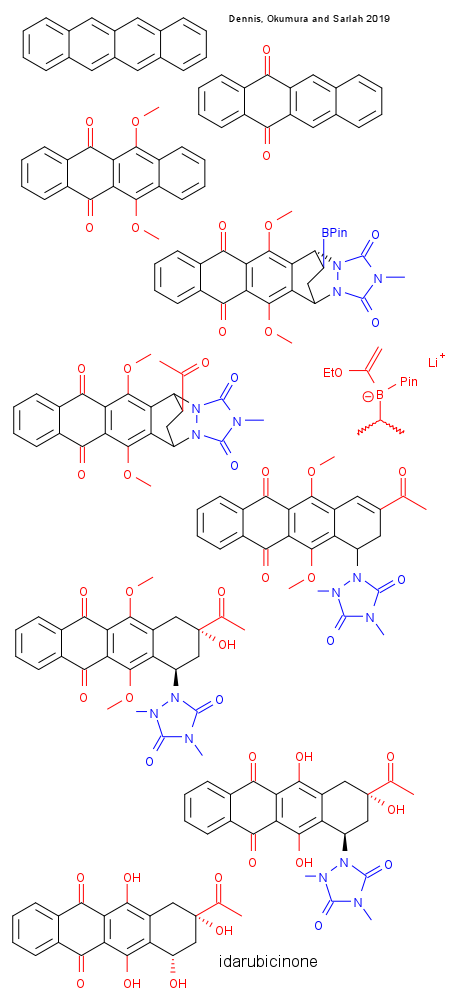 Dennis, Okumura and Sarlah report a synthesis of idarubicinone, a compound closely related to and precursor to Idarubicin, a well known antileukemic drug. (DOI) They are both members of the anthracycline class of drugs that work by intercalating with DNA. They can do that because they are flat as a plank and can fit nicely in the DNA helix. The source of the new idarubicinone synthesis is an interesting one: tetracene is known as an organic superconductor and not as a drug precursor but it shares with idarubicinone the flat 18 carbon framework. All it takes is 7 oxidation steps and two dearomatizations.
Dennis, Okumura and Sarlah report a synthesis of idarubicinone, a compound closely related to and precursor to Idarubicin, a well known antileukemic drug. (DOI) They are both members of the anthracycline class of drugs that work by intercalating with DNA. They can do that because they are flat as a plank and can fit nicely in the DNA helix. The source of the new idarubicinone synthesis is an interesting one: tetracene is known as an organic superconductor and not as a drug precursor but it shares with idarubicinone the flat 18 carbon framework. All it takes is 7 oxidation steps and two dearomatizations.
The main steps in summary: 1] oxidation by iodane phenyliodine(III) sulfate / cobalt(II) tetraphenylporphyrin 2] oxidation PIFA / (Cymene)ruthenium dichloride dimer (the only process found that was selective enough) 3 methylation with dimethyl sulfate / potassium carbonate 4] dearomatization photochemical reaction with N-methyl cousin of 4-Phenyl-1,2,4-triazole-3,5-dione , remarkably selective as this dienophile has two sides to choose from (not isolated) 5] hydroboration with catecholborane / tetrafluorborate cousin of Cyclooctadiene rhodium chloride dimer / dppb with subsequent replacement of unstable catechol with pinacol. Diastereoselective ratio 3:1 6] Ketone group protection Zinc, trimethylsilyl chloride 7 Zweifel olefination with 1-lithioethyl vinyl ether (to a boronate complex) then iodine / sodium methoxide Also de-masks the keto groups 8] Beta elimination of the urazole with methylation potassium tert-butoxide / dimethyl sulfate 9] Mukaiyama hydration PhSiH2(Oi-Pr)/ O2 // tris(dipivaloylmethanato)manganese 10] Methyl ether deprotection boron trifluoride 11] Elimination reaction of the urazole sodium dithionite / sodium hydroxide 12] Oxidation of intermediate double bond with oxygen.
