Perkins mauve reinvestigated
4 October 2008 - news (updated Oct. 19)
A Portuguese/English team has took it upon themselves to collect a bunch of Perkin mauve relics from several museums dated between 1856 and 1906, to put them through a series of chemical tests (DOI) with some surprising results. Several team members had in 2007 already identified a couple of new components in the Mauve mix (DOI) but now it turns out the dye consists of at least 13 derivatives of the same 7-amino-5-phenyl-3-(phenylamino)phenazin-5-ium core only differing with respect to methyl substitution pattern.
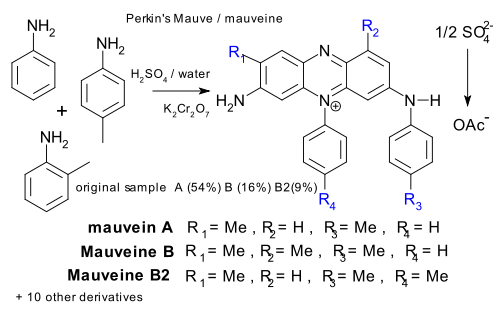
The new study also reveals two different production processes. In original samples (dated 1856 and onwards) the phenazinium cation is accompanied by a sulfate anion. This salt is amorphous and poorly water-soluble. In 1862 Perkin replaced sulfate by an acetate ion making the dye more soluble and to improve crystallinity he increased the amount of O-toluidine in the formulation.
interestingly a sample cherished by the Science Museum (Link) as the original stuff turned out to be post 1862 material. A Mauveine specimen also in possession by the science museum and now declared genuine was donated by Perkin's daughter (or granddaughter, the article is fuzzy at this point) in 1947.
Update: in another case of what we can call chemical archeology, samples from the famous 1953 Miller-Urey experiment have been re-analysed by Johnson et al. via HPLC / Time-of-flight mass spectroscopy at the sub-picomolar (<10-12 M) level revealing many more amino acids than Miller (obviously using less sophisticated equipment) himself had reported (DOI). This new finding however does not change the significance of the Miller experiments.
