Oseltamivir 61
08 July 2016 - Gone in 60 minutes
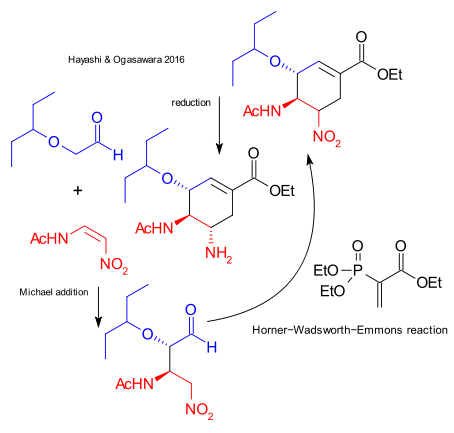 Is this really oseltamivir total synthesis number 61? We stopped counting the last time at number 27. Soon there will be a Journal of Oseltamivir Synthesis. Hayashi & Ogasawara present effort no 61 in Organic Letters (DOI) and Time Is Money! The opening paragraph contains two non-truths: "It is one of the goals for a chemist to synthesize a molecule as quickly as possible" and Tamiflu "is one of the most effective drugs for the treatment of influenza". But never mind, the big claim in this effort is a one-pot synthesis in just 60 minutes at the cost of an overall yield of just 15%. In a single reactor were added sequentially (the big picture): a nitroalkene and an aldehyde in an asymmetric Michael addition with reagents a proline / thiourea / formic acid catalyst system, an ethyl acrylate for an Horner-Wadsworth-Emmons reaction, TBAF for nitro group epimerisation and zinc (under microwave irradiation) for a final reduction.
Is this really oseltamivir total synthesis number 61? We stopped counting the last time at number 27. Soon there will be a Journal of Oseltamivir Synthesis. Hayashi & Ogasawara present effort no 61 in Organic Letters (DOI) and Time Is Money! The opening paragraph contains two non-truths: "It is one of the goals for a chemist to synthesize a molecule as quickly as possible" and Tamiflu "is one of the most effective drugs for the treatment of influenza". But never mind, the big claim in this effort is a one-pot synthesis in just 60 minutes at the cost of an overall yield of just 15%. In a single reactor were added sequentially (the big picture): a nitroalkene and an aldehyde in an asymmetric Michael addition with reagents a proline / thiourea / formic acid catalyst system, an ethyl acrylate for an Horner-Wadsworth-Emmons reaction, TBAF for nitro group epimerisation and zinc (under microwave irradiation) for a final reduction.
Note: The Just Like Cooking blog is suspicious.
