Novel palladium ArX-Y exchanges
29 September 2009 - catalysis
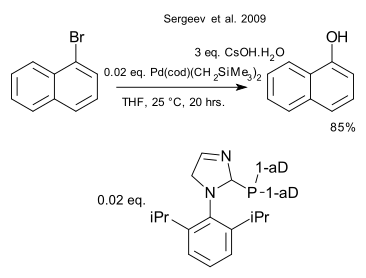
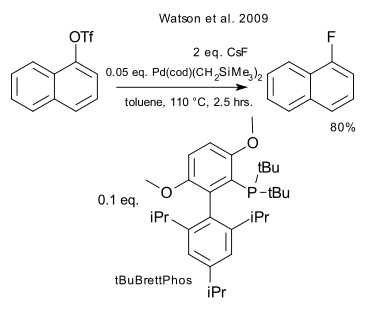
The two reactions depicted below have a lot in common: the substrate is an aryl halide or closely related triflate, the nucleophile is caesium fluoride or caesium hydroxide, the catalyst is based on palladium and the ligand is an organophosphine. Both reactions take place via a oxidative addition / metathesis / reductive elimination reaction mechanism.
The work done in the first set (Watson et al. DOI) by the Buchwald laboratory was inspired by earlier results obtained with reductive elimination of discrete R-Pd-F compounds. Key to the success for the formation of the organofluorine compound is the monomeric nature of the R-Pd-F/tBuBrettPhos intermediate.
Palladium catalysed hydroxylations have already been described in 2006 (Anderson et al. (again Buchwald lab) DOI) and 2007 (Chen et al. DOI) but the 2009 work done by Sergeev et al. ( DOI) takes the reaction to room temperature. As in the above reaction the R-Pd-X/ligand entity is a monomer and not (as is usual in organopalladium chemistry) a dimer.