Novel Imatinib base synthesis
19 Juli 2008 - Industrial organic chemistry
A new imatinib synthesis is presented as an alternative for the existing industrial method. But is it really an improvement and one suitable for industry?
Organic Process Research & Development (Link) is a scientific journal (ACS) dedicated to industrially relevant organic synthesis which is very different from regular academic laboratory synthesis. In industry and especially in pharma, reagents have to cheap, safe to handle and non-toxic, the reaction has to be reproducible and should be scalable. Side products and waste products have to be precisely known.
Hundreds of synthetic protocols are known en new ones invented every year for one and the same chemical transformation. Only those that have proven to be reliable will find their way into disciplines such as total synthesis (after all, why risk all yield with a fancy new reagent in step 25 of your synthesis) and even fewer end up in a chemical plant. This is why reading up on industrial synthesis is worthwhile if you want to filter out the relevant reactions from the wannabees.
As an example of research aimed at industrial production one involving imatinib. This cancer drug was one of the first offspring of rational drug design and if you believe the Wikipedia page hugely expensive despite its simple appearance (no stereocenters!). A group of Northwest University researchers set out to improve the existing Novartis procedure DOI and here is how they did it.
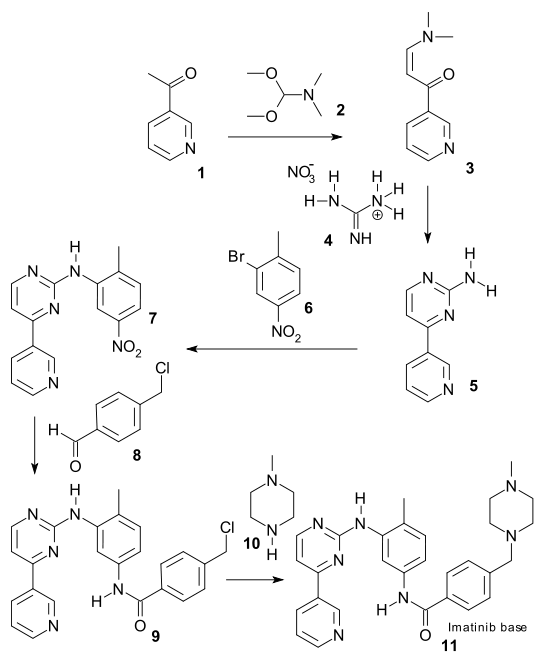
2-acetylpyridine (1) was alkylated with the acetal of N,N-dimethylformamide 2 to enamine 3. A pyrimidine ring in 5 was formed with base and reagent guanidine nitrate 4 and nitrotoluene fragment 6 was added in a Ullmann-type reaction with CuI generating secondary amine 7. The nitro group was reduced by hydrazine / FeCl3/C to the amine which was then converted to amide 8 with acid chloride 9. The final step is addition of piperazine 10 to form imatinib 11.
So is this procedure an improvement on the existing method and ready-made for industrial implementation? Surely they have eradicated the use of toxic cyanamide, cumbersome sodium metal and expensive palladium but they have also introduced equally toxic hydrazine and the harmful and explosive guanidine nitrate. As a further point of criticism the final step is demonstrated on a 0.5 gram scale. If the journal Organic Process Research & Development would live up to its standards the scale would at least be a kilogram.
Liu, Y., Wang, C., Bai, Y., Han, N., Jiao, J., Qi, X. (2008). A Facile Total Synthesis of Imatinib Base and Its Analogues. Organic Process Research & Development, 12(3), 490-495. DOI: 10.1021/op700270n
