New in transfer hydrogenation
07 September 2021 - Orgo
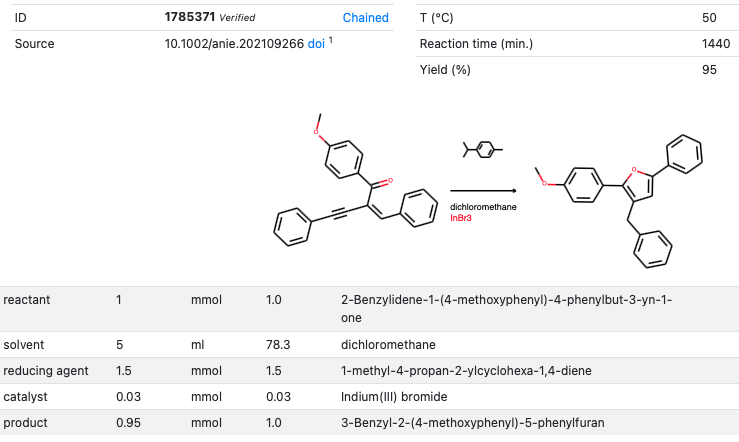 Kail, Weber and Hilt (Carl von Ossietzky Universität Oldenburg) report a new transfer hydrogenation reagent in a recent Angewandte publication (DOI). Hydrogen gas is the obvious choice in hydrogenation reactions but alternative hydrogen donor compounds exist such as diazene and 1,4-cyclohexadiene.
Kail, Weber and Hilt (Carl von Ossietzky Universität Oldenburg) report a new transfer hydrogenation reagent in a recent Angewandte publication (DOI). Hydrogen gas is the obvious choice in hydrogenation reactions but alternative hydrogen donor compounds exist such as diazene and 1,4-cyclohexadiene.
The new reagent, 1-methyl-4-propan-2-ylcyclohexa-1,4-diene (gamma-terpinene) was tested against certain alkynyl enones to form furan compounds. Indium tribromide was found to be an effective catalyst. The article describes how the reaction was optimized using a Design of Experiments algorithm that allowed the optimization of 6 reaction variables (catalyst loading, reaction time, substrate concentration, amount of reducing agent, temperature and type of reducing agent) with just 16 experiments. Within this context it was also found that catalyst loading had little impact compared to the reducing agent loading.
As to the reaction mechanism, the authors propose (by deuterium labeling experiments) that the indium Lewis acid first activates the keto group and then coordinates to the alkyne group. A second equivalent of indium tribromide accepts a hydride ion from the reducing agent converting it into the corresponding Wheland complex. The hydride then adds to the enone in a Michael addition and then the cyclization reaction is triggered. In the final step the Wheland complex donates a proton.
