New in metathesis
10 June 2017 - Orgo
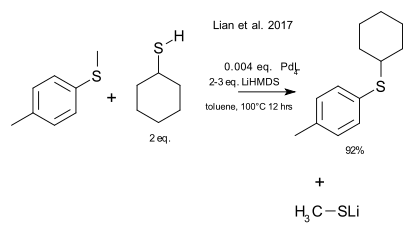 Chemists have been mapping out the scope of the metathesis reaction for over 60 years now and with the availability of many more elements than just carbon the job is not done yet. Lian, Bhawal and Morandi report on an aryl alkyl sulfide - thioalkyl variation in Science. (DOI)
Chemists have been mapping out the scope of the metathesis reaction for over 60 years now and with the availability of many more elements than just carbon the job is not done yet. Lian, Bhawal and Morandi report on an aryl alkyl sulfide - thioalkyl variation in Science. (DOI)
This not exactly the olefin metathesis chemistry most people are familiar with but more a metathesis in the sense of an exchange reaction. As a typical reaction hexanethiol was found to react with methyl p-tolyl sulfide to have the methyl group exchanged with the cyclohexyl group. This reaction required an NHC type palladium catalyst and LiHMDS. In terms of reaction mechanism it is a ligand exchange with an oxidative addition and reductive elimination. As an added bonus it was found that commercial polymer polyphenylene sulfide could be depolymerised in similar reaction conditions.
Rik
