Nanotubes unzipped
03 May 2009 - Nanotechnology
Two back-to-back articles in the April 16 issue of Nature report on how to get nanoribbon from nanotubes. Nanotubes are the cylinders of chicken-wire hexagons of carbon and nanoribbons are the graphene strips so making one using the other makes sense. Graphene nanoribbons have been produced before by lithographic patterning of graphene sheets, by chemical vapor deposition and by sonification of graphite.
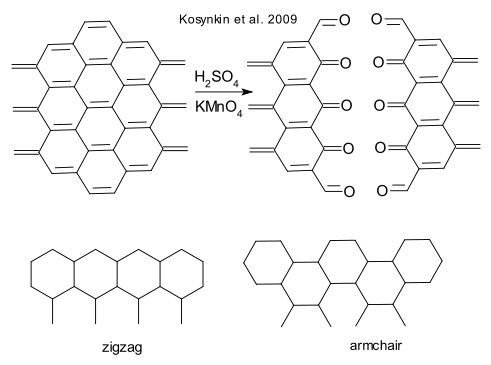
A group around James M. Tour (of nanocar fame) has successfully cut open a multiwalled nanotube lengthwise in a sulfuric acid / potassium permanganate solution in water (Kosynkin et al. DOI). The permangate oxidizes the double bonds in the nanotube just as in regular alkenes and the newly formed nanoribbons have the edges all oxygenated by ketone groups. Reduction was accomplished with hydrazine and ammonium hydroxide and a surfactant. The nanoribbons (yield > 90%) are difficult to work with, they easily fold on to itself (like adhesive tape) and tend to aggregate. It is was not possible to distinguish between zigzag edges or so-called armchair edges.
In the second article, Jiao et al. (DOI) take another approach and simply etch away the top from a nanotube by argon plasma etching. They do this in several steps: deposition of MWCNT material on silicon, spin-coating a layer of PMMA on top of it (partially submerging and protecting the tubes), peeling away the film thus obtained and apply the plasma. The top half is basically destroyed, reducing the yield to 20%.
These are two successful methods for the unzipping of nanotubes but should also be applicable to fullerenes. Some effort has already been spent in opening up fullerenes for example see Endohedral hydrogen fullerene.
