Deoxygenation unexplained
18 August 2008 updated 20 August
What should be a straightforward exercise in beta-aminoketone synthesis by several Chinese researchers (Yong Xin et al. 2008 DOI) ends up in mayhem. This info-graphic sums it all up:

Salicylaldehyde and the trimethyl silyl ether of cyclohexanone are reacted in presence of boron trifluoride expecting a double Mukaiyama aldol addition accompanied by acetal formation and condensation. Surprisingly at the end of the road one of the alkene groups is hydrogenated or as you like one of the hydroxyl groups is deoxygenated. When the ketone ring size is reduced this happens twice but the expanded ring on the other hand gives the expected product.
According to a 2006 publication ( DOI) this research group has had to face unexpected results before.
In trying to solve this mystery the experimental details are not always helpful. In the text the reaction takes place in benzene but the general procedure prescribes dichloromethane, compound labels are mixed up, the exact excess of aldehyde is ill-defined and elemental analysis for compound C lacks a complete carbon atom. The chemical yield for B is low but details on the how&why are missing.
The authors refuse flat-out to speculate about how this reaction is supposed to take place and kindly invite us to read a forthcoming paper. Surely there must be some literature out there on the use of BF3 on deoxygenations? In fact the literature offers numerous examples of reductions by this reagent in combination with a hydride donor such as triethylsilane. One report (Smonouet et al. 1988 DOI) shows how 1-phenylethanol is converted to ethylbenzene in this way with formation of a phenethyl carbocation as a rate-determining step.
Ring-strain no doubt disfavors the elimination reaction in the smaller cycle and the only thing missing in the present reaction then is the nature of the hydride donor. Three equivalent of water are generated in the reaction so that is a start.
And surely at some point in the 200 years of chemical endeavor someone must have come up with the idea to mix cyclohexanone and salicylaldehyde just for the heck of it. Surprisingly, CAS at least throws up zero results when queried. Only in one instance (check up details in the Azerbaidzhanskii Khimicheskii, page 6, 1978 if you have a copy) a salicylaldehyde derivative is given a chance to meet up with cyclohexanone.
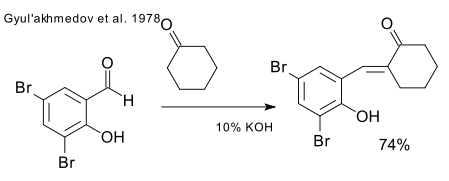
For the only mention of the direct cyclohexanone-salicylaldehyde adduct please consult the equally informative Zhurnal Organicheskoi Khimii (1988, page 2419).
